Central Biobank Magdeburg
 Research into the development, treatment and prevention of diseases is indispensable for the development of preventive measures, improved diagnostic options and new treatment approaches. The essential foundation of biomedical research is the collection and use of human samples and disease data. In recent decades, biomaterial banks have developed into an independent component of modern medical research due to justified and growing demands regarding sample quality and data protection and the increasing networking of interinstitutional medical research.
Research into the development, treatment and prevention of diseases is indispensable for the development of preventive measures, improved diagnostic options and new treatment approaches. The essential foundation of biomedical research is the collection and use of human samples and disease data. In recent decades, biomaterial banks have developed into an independent component of modern medical research due to justified and growing demands regarding sample quality and data protection and the increasing networking of interinstitutional medical research.
Biobanks are in demand internationally and by third party funding providers as a “key resource” used as a long-term tool and as an important location factor for biomedical research. In addition to the quality-assured storage, processing and distribution of samples and sample-related data, the tasks assigned to a biobank include compliance with data protection and ethical framework conditions as well as the maintenance of quality-assured concepts for the use of biomaterials and associated data.
The following institutes are directly involved in the establishment and operation of the biobank:
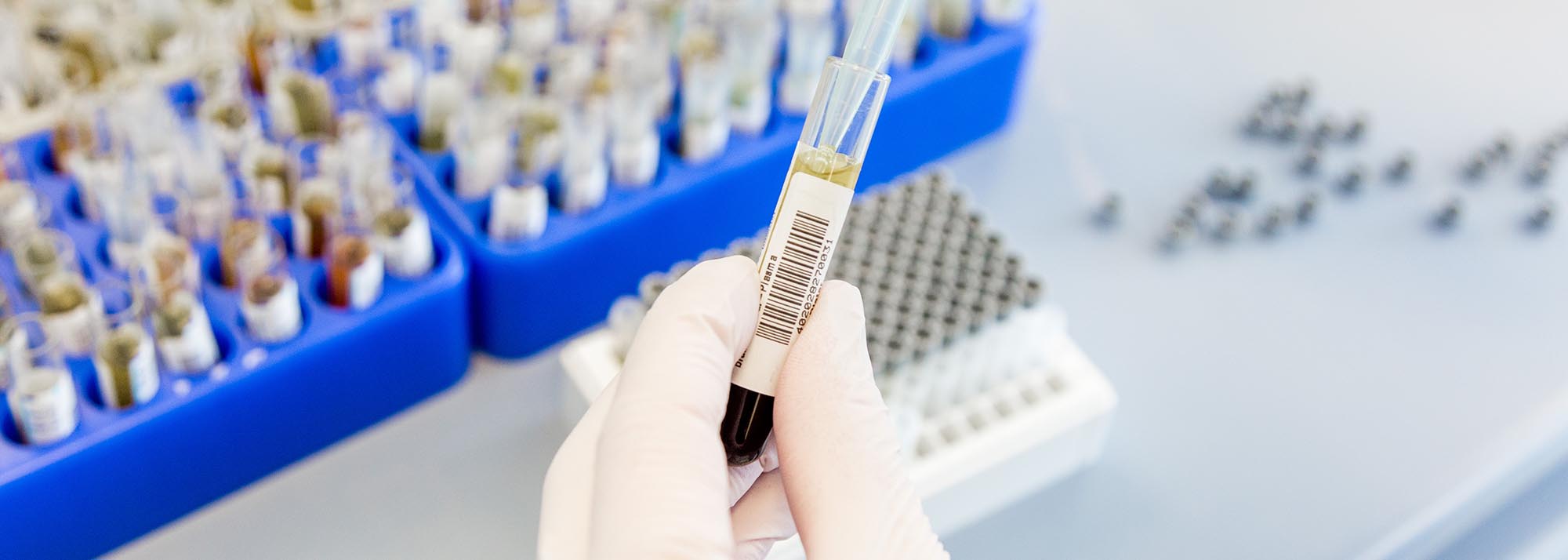
- Institute for Clinical Chemistry and Pathobiochemistry (IKCP) – Storage of liquid samples
- Institute for Pathology (IPA) – Storage of tissue samples
- Institute of Biometry and Medical Informatics (IBMI) – Data analysis
The Medical Computer Center (MRZ) supports the biobank in the area of data management and IT.

The establishment of the central biobank Magdeburg (ZBM) is mainly supported by the European Union within the framework of the European Regional Development Fund.
The ongoing operation is to be financed by a scale of fees.
Services provided by the Biobank:
- Research funding through the provision of samples and data
- Support in the acquisition of third-party funds
- Optimal use of existing structures
- Economy à Cost-covering operation
- Public Relations: Homepage UMMD
- Workflow design for users
- Administration of patient consent "Broad Informed Consent", data protection
- Documentation: Transparency and traceability
- Administration of sample data, patient data, analysis data + data mining
- Quality assurance: SPREC, cooling system monitoring, sample metadata